|
Host factors are required at every step in this life cycle and represent candidate druggable targets (i.e., host-targeting antivirals) with the potential for broad-spectrum activity against multiple viruses within a given virus family and even across virus families. Coronaviruses replicate via a well-established series of molecular events. Broad-spectrum antivirals could also reduce morbidity associated with common cold-causing HCoVs including OC43, NL63, 229E, and HKU1.Īntivirals segregate into two basic categories, virus-targeting and host-targeting, both of which require an understanding of the molecular mechanisms used by viruses to replicate in host cells. Moreover, considering that SARS-CoV-2 is the third novel human coronavirus (HCoV) to emerge and cause serious disease in the human population in the past two decades following SARS-CoV and MERS-CoV, potent and broad-spectrum antivirals will leave us better prepared to deal with future pandemics. 
While the importance of this scientific achievement cannot be overstated, there is still a great need for novel antiviral therapies for use in vulnerable immunocompromised individuals, in regions where vaccine access is limited, and in the event antigenically distinct SARS-CoV-2 variants, such as delta and omicron, continue to arise and threaten vaccine efficacy. Multiple COVID-19 vaccines recently demonstrated high efficacy, received FDA approval, and are being administered to people across the globe. The causative agent of COVID-19, SARS-CoV-2, spread quickly across the planet resulting in 245 million infections and nearly 5 million deaths at the time of this writing. The COVID-19 pandemic is arguably the most consequential infectious disease outbreak in modern times. Overall, our studies substantiate and expand the growing body of literature focused on understanding key human coronavirus-host cell interactions and exploit that knowledge for rational antiviral drug development. Finally, we identified novel candidate antiviral compounds targeting a number of factors revealed by our screens. The role of EDC4 and XRN1 in coronavirus replication in human small airway epithelial cells was verified. Of note, host factors involved in cell cycle regulation were enriched in our screens as were several key components of the programmed mRNA decay pathway. The website was created to allow visualization and comparison of SARS-CoV2 CRISPR screens in a uniformly analyzed way. While we identified multiple genes and functional pathways that have been previously reported to promote human coronavirus replication, we also identified a substantial number of novel genes and pathways. 
Gene knockdown, CRISPR knockout, and small molecule testing in Vero, HEK293, and human small airway epithelial cells were used to verify our findings. To identify host factors that promote SARS-CoV-2 infection with potential for broad-spectrum activity across the coronavirus family, we performed genome-scale CRISPR knockout screens in two cell lines (Vero E6 and HEK293T ectopically expressing ACE2) with SARS-CoV-2 and the common cold-causing human coronavirus OC43. Viruses require host factors at every step in their life cycle, representing a rich pool of candidate targets for antiviral drug design. While effective vaccines are being administered globally, there is still a great need for antiviral therapies as antigenically novel SARS-CoV-2 variants continue to emerge across the globe. 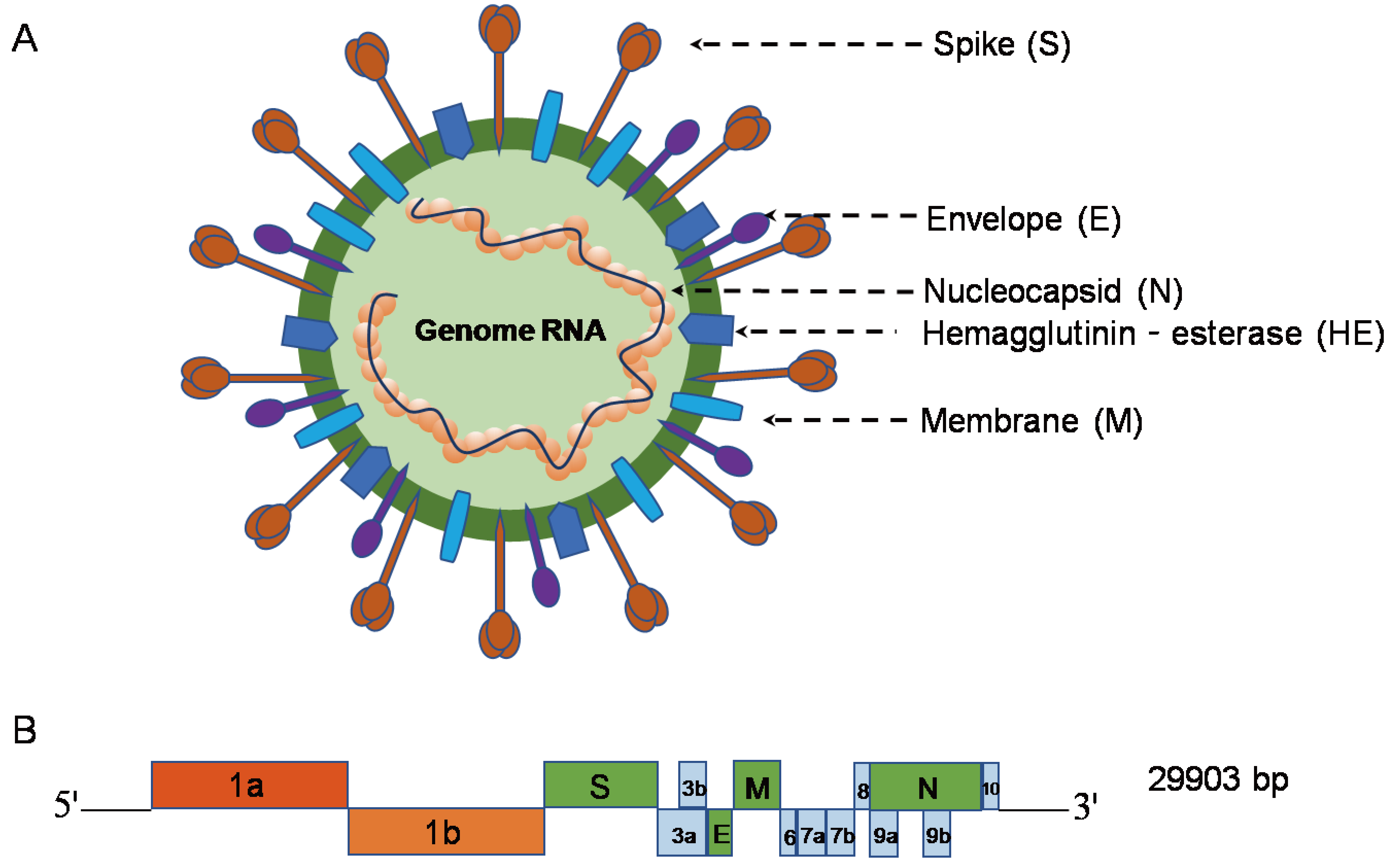
The COVID-19 pandemic has resulted in 275 million infections and 5.4 million deaths as of December 2021.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
